Veregen®
SB03, also known as Veregen® (sinecatechins), 10% is an ointment indicated for the topical treatment of external genital and perianal warts (EGW) in patients 18 years and older. These warts are caused by the human papillomavirus (HPV) infection.
Veregen® is the first botanical drug approved for prescription use in the U.S. The NDA of Veregen® was approved by TFDA in March, 2013.
- Natural and Safe
Contain catechins mixture, extracted from green tea leaves. With very good safety profile, systemic side effects are unlikely.
- Good Efficacy
High complete clearance rate in baseline and newly emerging warts.
- Lowest Recurrence Rate
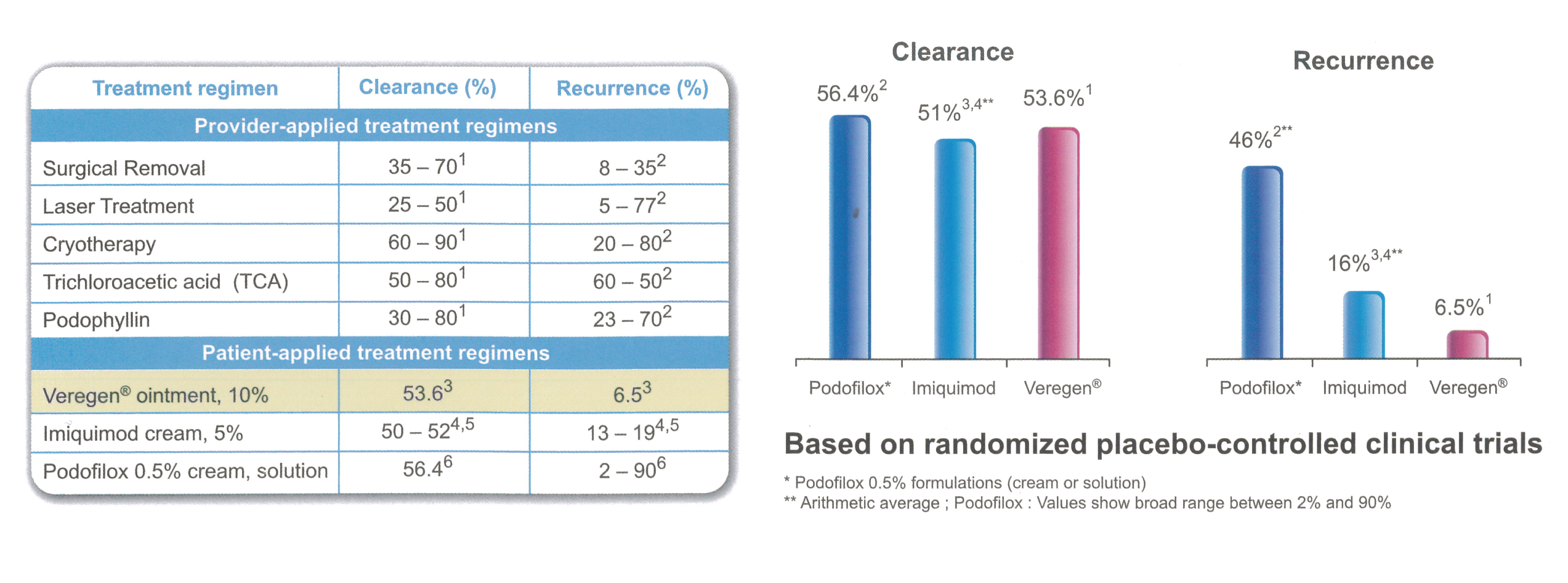
- Easy Patient-Applied Administration
No drug holiday
No wash off
More information please find the News or the website for MediGene (German).
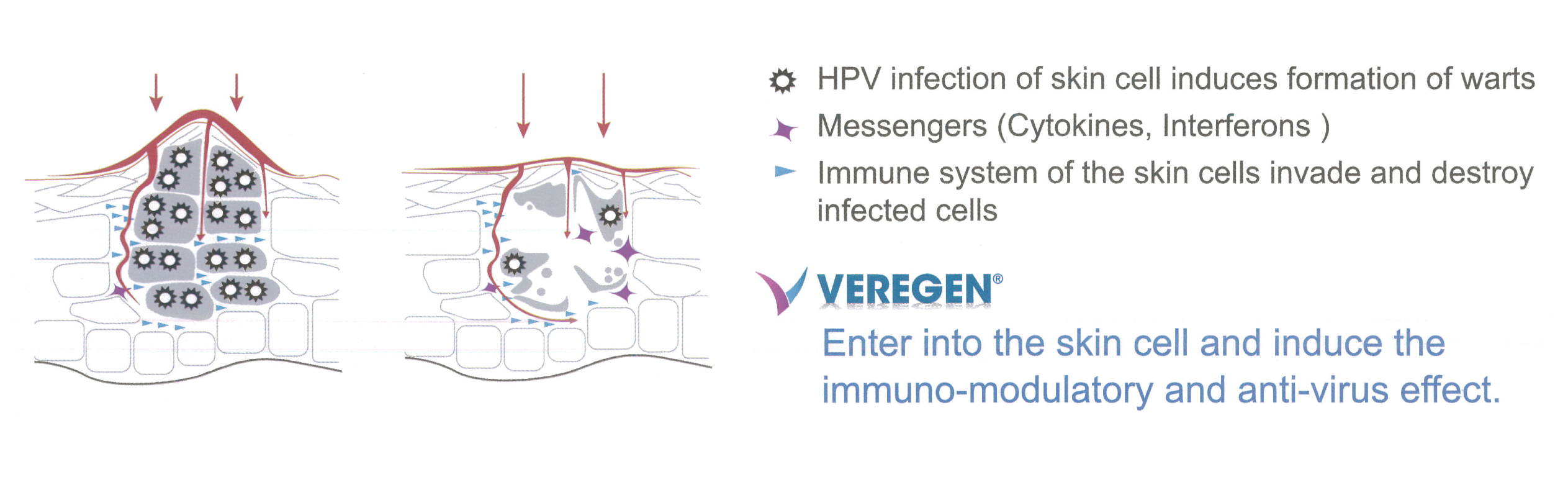
Veregen® MOA
Veregen® Worldwide